DOUBLED PFS VS 5-FU/LV ALONE1,7*

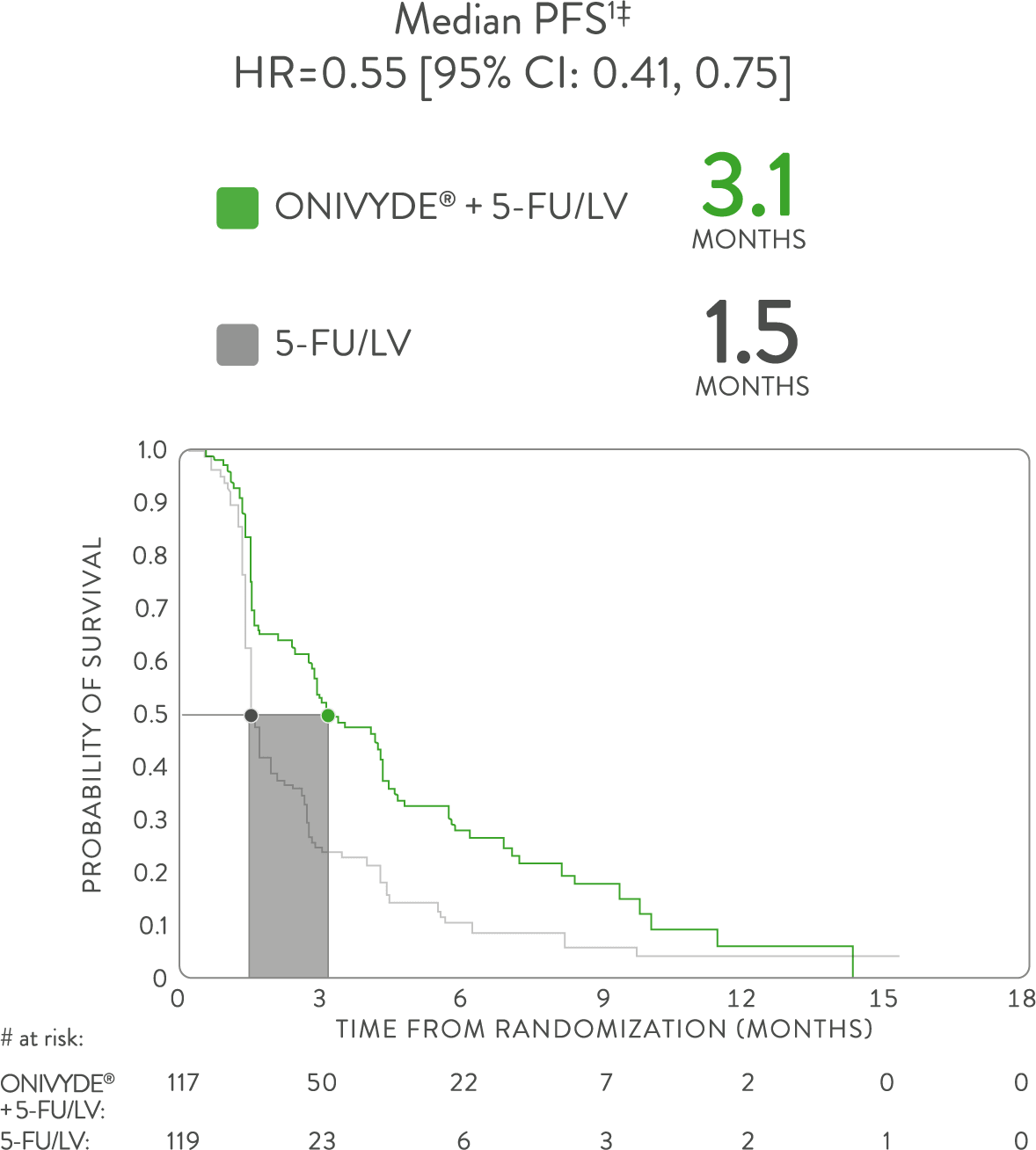
ONIVYDE + 5-FU/LV doubled PFS compared with 5-FU/LV alone, in the NAPOLI-1 primary analysis1
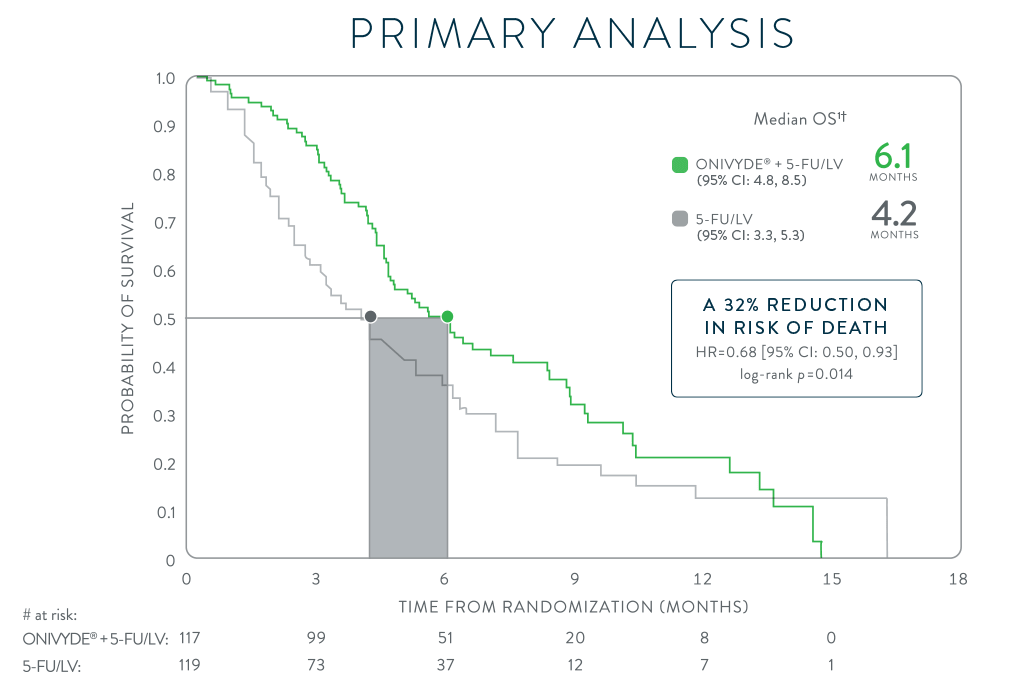
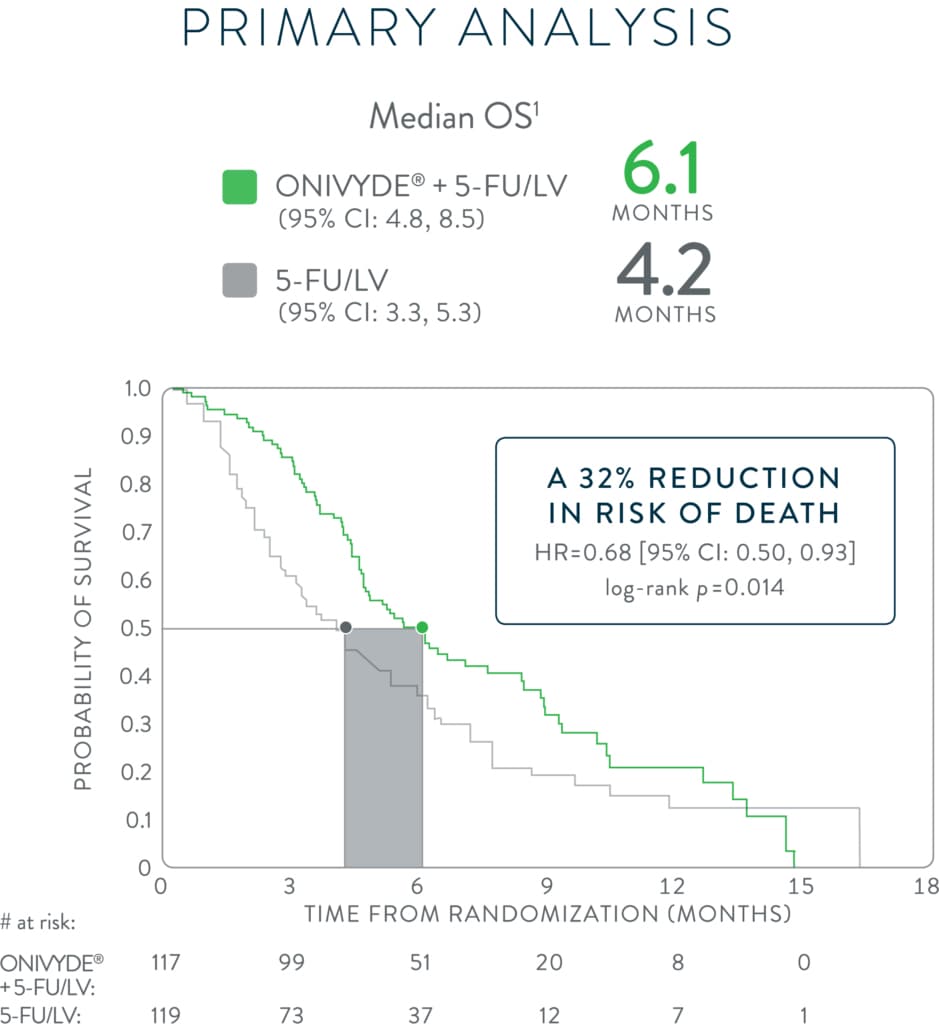
*NAPOLI-1 was a global, phase 3, randomized, open-label, multicenter trial in patients (N=417) with metastatic adenocarcinoma of the pancreas whose disease had progressed following gemcitabine-based therapy. Patients were initially randomized to receive ONIVYDE (100 mg/m2 every 3 weeks) or 5-FU/LV. After 63 patients were enrolled, a third arm, ONIVYDE (70 mg/m2 every 2 weeks) + 5-FU/LV, was added. Treatment was continued until disease progression or unacceptable toxicity. The primary endpoint, median OS, was assessed with 2 pair-wise comparisons: ONIVYDE (n=151) vs 5-FU/LV (n=149) and ONIVYDE + 5-FU/LV (n=117) vs 5-FU/LV (n=119, post-protocol amendment). There was no improvement in OS for ONIVYDE vs 5-FU/LV (HR=1.00, p=0.97 [2-sided log-rank]).1,7
HR=hazard ratio; CI=confidence interval; 5-FU=fluorouracil; LV=leucovorin.

Confirmed objective response rate (ORR) in NAPOLI-1
- ONIVYDE + 5-FU/LV demonstrated a confirmed ORR of 7.7% (9/117) and 5-FU/LV demonstrated a confirmed ORR of 0.8% (1/119)1†
NAPOLI-1 was a global, phase 3, randomized, open-label, multicenter trial in patients (N=417) with metastatic adenocarcinoma of the pancreas whose disease had progressed following gemcitabine-based therapy. Patients were initially randomized to receive ONIVYDE (100 mg/m2 every 3 weeks) or 5-FU/LV. After 63 patients were enrolled, a third arm, ONIVYDE (70 mg/m2 every 2 weeks) + 5-FU/LV, was added. Treatment was continued until disease progression or unacceptable toxicity. The primary endpoint was median OS. Additional efficacy endpoints were PFS and ORR.1,7
†Investigator assessment per RECIST v1.1.7
RECIST=response evaluation criteria in solid tumors.